|
10/26/2022 0 Comments Electron affinity Infringement Notice, it will make a good faith attempt to contact the party that made such content available by If Varsity Tutors takes action in response to Information described below to the designated agent listed below. Or more of your copyrights, please notify us by providing a written notice (“Infringement Notice”) containing If you believe that content available by means of the Website (as defined in our Terms of Service) infringes one 
When all the dipole moments of polar bonds in a molecule are summed, the molecular dipole moment results, as per the following equation.ĭipole moment = charge * separation distance This leads to the O-H bond having a dipole moment. In other words, oxygen more strongly attracts electrons when in a bond with hydrogen. 
Electronegativity is a property inherent to the atom in question, whereas dipole moment is a property of the bond between them.įor example, oxygen has an electronegativity of 3.44, and hydrogen of 2.20. Dipole moment, however, is dependent on the electronegativity of the atoms making up the bond. When comparing more than one polar covalent molecule, we use the dipole moment value to help us determine relative strength of polarity. 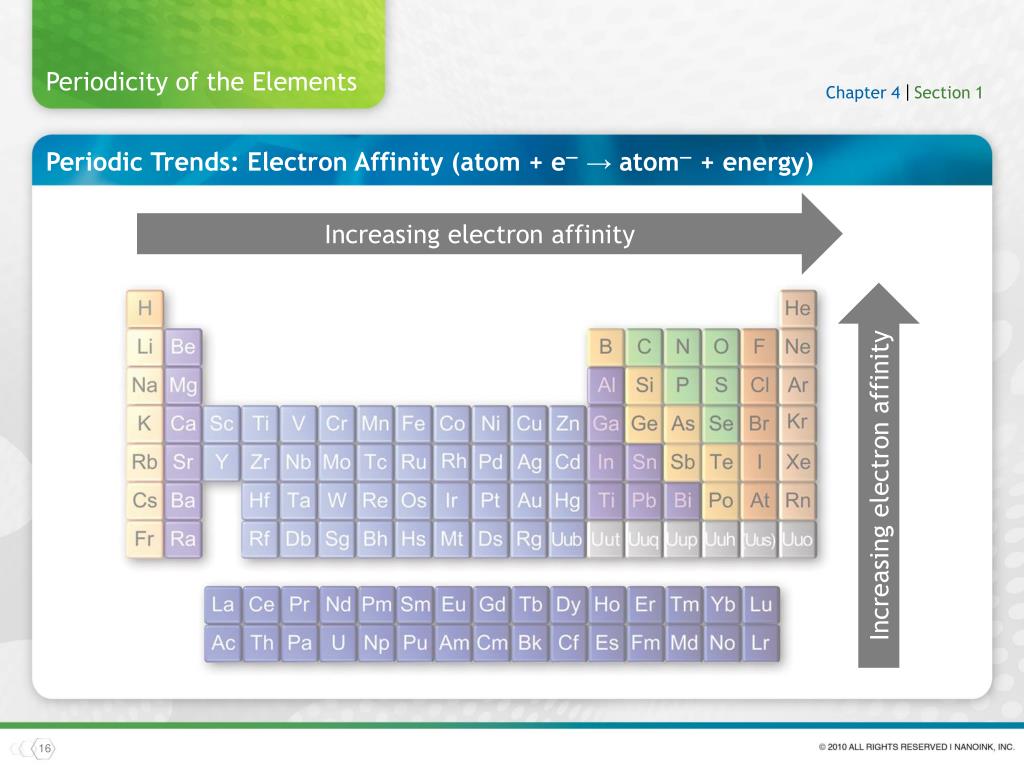
The former is purely ionic, and the latter is polar covalent. An example can be seen by contrasting sodium chloride, NaCl, with an organic molecule, R-C-OH. Polar compounds are different from those compounds that are purely nonpolar or purely ionic. Electronegativity is an important concept in physical chemistry, and often used to help quantify the dipole moment of polar compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
